Treating Discogenic Back Pain with Bone Marrow Concentrate is Superior to Lumbar Disc Replacement at Two-Year Follow-up
Kenneth A. Pettine D. Maxwell
DOI : https://doi.org/10.31546/JBRCI.1002
Download Article : Peer-reviewed Article PDF
Abstract
Objective: Millions of patients suffer chronic back pain from degenerated lumbar discs. Surgical options for one or two levels of discogenic back pain are fusion or lumbar artificial disc replacement [LADR]. Study Design: This study compares the complications, reoperations, cost, and clinical outcomes of bone marrow concentrate [BMC] versus LADR for the treatment of discogenic back pain. The BMC study is prospective non-randomized class 2 data. The LADR studies were prospective randomized class 1 data. All operations were performed at the same private clinic. All studies had IRB approval. Follow up on all the studies was two years. Methods: The BMC was a 30-minute procedure involving aspiration of 55ml of bone marrow from the iliac wing, concentrating via centrifugation to a volume of 6ml and then injecting approximately 2ml of bone marrow concentrate per symptomatic lumbar disc. Clinical success in the LADR and BMC studies required a 50% improvement in ODI and VAS from pre-operative through two-year follow-up. Results: Clinical failure of LADR occurred in 27% (24 of 89 patients) at two-year follow-up. Five patients in the BMC study had surgery within two years of the BMC injection and were considered failures (19%). The remaining 21 BMC patients improved an average of 64% in ODI and 72% in VAS. Conclusions: These results indicate a BMC injection may be a reasonable non-surgical option for patients with symptomatic discogenic back pain.
Keywords: Mesenchymal Stem Cells; Stem cells; Cell-based therapy; Bone Marrow Concentrate, Lumbar Spine, Disc Replacement.
Introduction
Direct and indirect costs of discogenic back pain in the United States exceeds 100 billion dollars annually [1,2]. Non-operative treatments include weight loss, exercise, heat, ice, NSAIDs, chiropractic, physical therapy, acupuncture, oral steroids, steroid injections, and opiates [3,4,5]. The use of opiates to treat chronic low back pain is a major contributor to the current opioid crisis [3,4].
Surgical options for one or two levels of discogenic back pain are fusion or lumbar artificial disc replacement (LADR) [22,23]. LADR was developed to potentially improve on the clinical results of lumbar fusion for treating discogenic back pain and reduce the development of adjacent level disc pathology resulting in repeat fusion surgery [12,13,14]. Numerous prospective randomized studies have reported superior but non-significant improvements in VAS and Oswestry Disability Index (ODI) comparing LADR to fusion [15,16,17,19,20,24,25,26,28].
The use of biologics to treat disc abnormalities is a possible non-surgical option which potentially can bridge the gap between traditional non-surgical treatments for discogenic back pain and surgery. There is mounting evidence to support the use of biologic and cell-based therapy for chronic discogenic low back pain. The authors of this paper have published one, two, three and five- year follow-up from a study assessing the safety and efficacy of BMC cells as an alternative to surgery for discogenic back pain at one or two levels [8,9,10]. The authors have also published the results of treating multi-level discogenic back pain with BMC.32 There have been numerous studies utilizing mesenchymal stem cells to enhance tissue repair and decrease inflammatory damage in both in vitro lab studies and in vivo clinical models. It is known that BMC, the treatment used in this study, contains mesenchymal stem cells as well as a number of other cell types including but not limited to: hematopoietic stem cells, endothelial progenitor cells, and platelets. Studies have shown both the mesenchymal stem cell population and other nucleated cell types have healing properties and may contribute synergistically to the healing seen in studies on the spine [18,19,20,21,27].
This is the first study to compare the clinical results of intradiscal BMC to LADR for the treatment of patients who have discogenic back pain.
Materials and Methods
Study Design
This study is a prospective non-randomized evaluation of patients having an injection of BMC (class two data) compared to LADR. All studies had IRB approval. The LADR studies were prospective randomized studies (class one data). Follow up in all the studies was two years. The patients enrolled as subjects in all these studies presented clinically with symptomatic moderate to severe chronic axial back pain. Abnormalities were present on lumbar MRI scanning and plain radiographs. These abnormalities include anterior and posterior osteophyte formation, disc space narrowing on plain radiographs, and nucleus pulposus desiccation on MRI scanning.
In all the studies pre-treatment disability (ODI) had to have a minimum of 30mm/100mm and pre-treatment baseline axial back pain had to be at least 40mm/100mm on visual analog scale (VAS) pain scores to meet inclusion criteria. The patients were required to sign and fully comprehend an informed consent document before participating in the study. All patients underwent a pre-injection medical history and physical examination along with the ODI and VAS pain scores. These questionnaires were repeated at six weeks, three months, six months, 12 months, and 24 months post injection of BMC or LADR. The patients’ primary physical complaint in the BMC study was one of axial central lumbar pain without leg pain. Standard exclusion criteria for the BMC study included evidence of a symptomatic herniated disc. The IDE studies accepted patients with a herniated disc and associated leg pain.
The inclusion/exclusion requirements were similar in both IDE studies.
Definition of Clinical Success
The definition of clinical success in all the LADR and BMC studies, for the basis of this paper, required a 50% improvement in ODI and VAS from pre-operative through two-year follow-up. This is more stringent than the 15-point improvement required for the FDA IDE trials. In addition, any patient undergoing a reoperation or showing signs of neurologic deterioration were considered a failure. A 50% improvement in ODI and VAS was felt to be a reasonable stringent definition of success. Glassman, et al. published a study determining this was the improvement level where patients perceive clinical success [28].
Materials and Methods for BMC Study
There were 13 one level and 13 two level BMC procedures. BMC patient demographics are listed in Table 1.
Bone Marrow Collection and Processing
Bone marrow aspirate (BMA, 55ml) was collected over acid citrate dextrose-anticoagulant (ACD-A, 5ml) from the patient’s posterior iliac crest. The procedure was performed with IV sedation consisting of Versed and Fentanyl. Fluoroscopy confirmed the positioning of the Jamshidi needle in the iliac wing. BMA was collected in a 60ml syringe in a series of discrete pulls on the plunger (targeting a collection of 5-10ml per pull) with repositioning of the needle tip between pulls based on the reported enrichment of progenitor cells by (Hernigou et al. 2013) [4,5]. The BMA was processed using the ART21 system (Celling Biosciences, Austin, TX) to produce a bone marrow concentrated cell preparation. The 55ml of BMA were centrifuged for 12 minutes to produce 6ml of BMC. The 3ml of BMC were drawn from the processed device, and then 0.175cc of 50% glucose and 0.175cc of bicarbonate were added to the 6ml of BMC which was then immediately transferred to the physician for injection.
Packs and shipped overnight to the cell analysis laboratory (Celling Biosciences, Austin, TX). The samples were received and processed immediately to determine total nucleated cell (TNC) count and viability using a Nucleo-Counter NC-100 (Chemo-metric, Denmark). The BMC was diluted in phosphate buffered saline (PBS, Invitrogen, Grand Island, NY) with 2% fetal bovine serum (FBS, HyClone human mesenchymal grade, Thermo Scientific, Waltham, MA) and subjected to a Ficoll-Paque (GE Healthcare Life Sciences, Piscataway, NJ) gradient separation (1:1 cell solution to Ficoll ratio by volume) in order to deplete red blood cells. Analysis of the recovered cells included performing colony-forming unit-fibroblast and osteogenic (CFU-F and CFU-O, respectively) assays and phenotypic analysis by flow cytometry. For phenotype analysis, fresh (non-cultured) BMC cells were stained with a series of rabbit anti-human monoclonal antibodies for a hematopoietic lineage-committed (non-progenitor) panel of markers including CD2, 3, 8, and 11b (APC-Cy7), CD34 (PE), CD90 (FITC), and CD105 (APC) as well as appropriate isotype controls. Isotype, single color stain, and four-color stain samples were analyzed by a Guava Easy-Cyte 8HT (Millipore, Billerica, MA). The CFU-F assay was performed by creating a dilution series (in culture medium with 5% FBS and 1% antibiotics) of each cell preparation at concentrations of 50,000-500,000 TNC per well in standard 12-well plates. The plates were placed in an incubator at 37°C, 5% CO2, and 100% humidity for 72 hours when the medium was replaced. Medium was replaced every three days. After nine days in culture, wells were gently washed with PBS, fixing the colonies/cells with methanol, staining the attached cells with Crystal Violet, rinsing with water, and air-drying the plates. Visualization and counting of the colonies were done with an inverted microscope. Colonies containing 20 or more cells were scored as a CFU-F. The CFU-O assay was performed identically as CFU-F, but after nine days the medium was changed to an osteogenic induction medium (Advance STEM Osteogenic Differentiation Kit, Hy-Clone, Logna, UT) for an additional nine days with complete medium change every three days. On day 18, the wells were washed with PBS, then fixed for 15 minutes in 2% formalin solution, and contained for alkaline phosphatase activity (Vector Blue ALP, Vector Labs, Burlingame, CA) and calcified extracellular matrix (0.5% Alizarin Red solution, Sigma-Aldrich, St. Louis, MO).
Statistical Analysis in the BMC Studies:
Patient follow up outcomes were obtained by independent reviewers who were not investigators with the study. The reviewers were senior pre-med students. Uni-variable data comparisons of baseline to follow-up were analyzed using a two-tailed student’s t-test with a 95% confidence interval (Alpha=0.05, Microsoft Excel).
Intradiscal Injection
With the patient in a prone position, the injection site(s) was treated with local anesthetic (1% buffered lidocaine). BMC was percutaneously injected into the symptomatic disc(s) through a standard posterolateral discogram approach with a two-needle technique. The injection point of the 22-gauge needle was verified with fluoroscopy without use of contrast agent. Approximately 2-3 ml of BMC were used per symptomatic lumbar disc injection. Patients were prescribed pain medicine to be used as needed for three days and put on restricted physical activity for two weeks.
Materials and Methods of the LADR studies
These studies involved an analysis of every patient in an FDA IDE study undergoing a LADR at a single FDA IDE site. Every patient was part of two different FDA IDE studies. The first study involved comparing the results of the Maverick LADR versus LT cage with BMP. The second FDA study involved a comparison of the Charité versus Kineflex LADR. All of these studies were one level surgeries.
These 91 FDA IDE patients from two studies provide the basis for this paper. All the BMC and LADR cases were consecutively performed by the same private practice in the same ambulatory surgery center. The follow up at 2 years was 96% in the FDA studies. Patient demographics are listed in Table 2.
Facet Pain
Facet pain was diagnosed in patients who complained of continued pain after LADR. These patients typically had pain worse with extension and often unilateral off midline. These patients underwent intra-articular injection of the facet joint under fluoroscopic control. Each facet joint was anesthetized with 0.25ml of 0.25% Marcaine and 0.25ml of Depo-Medrol. These patients required at least an 80% improvement in their symptoms following this injection. In those patients achieving at least an 80% improvement in their symptoms, after the first facet injection. If their pain returned, a second injection was performed. Again, if they achieved an 80% reduction in their pain, they were considered to have pain secondary to facet pathology.
Results of LADR Studies
Based on a strict definition requiring at least a 50% improvement in ODI and VAS at two-year follow-up with no reoperations or deterioration in neurologic status, overall clinical failure occurred in 26% (24 of 91 patients) at two-year follow-up. Clinical failure occurred in 28% (7 of 25) of the Maverick patients, 39% (12 of 31) of the Charité patients and 14% (5 of 35) of the Kineflex patients. This is explained in Table 3.
Cause of Failure
Facet pathology was the etiology of failure in 50% (12 of 24) of the failure patients. All of these patients were diagnosed as having facet pain based on two separate injections as defined in the methods section. Nine of these patients went on to have percutaneous facet rhizotomy performed. None of these 12 patients have gone on to any additional surgery. Presently, they are living with their symptoms. This is explained in Table 4.
Implant complications occurred in five of the total 91 patients. These five patients had the following complications. One Maverick patient became infected 18 months after the initial surgery requiring surgical debridement and fusion. One Charité patient underwent revision of the implant within 24 hours post initial operation to a more midline position. One Charité patient underwent a posterior fusion secondary to bilateral pedicle fractures occurring six weeks post operatively. One Charité patient underwent a posterior fusion secondary to an implant invaginating into the L4 vertebral body at six weeks post op. One Kineflex patient subluxed the sacral endplate anteriorly approximately 4mm and underwent conversion to an anterior lumbar interbody fusion within 24 hours of the index operation. This is explained in Table 5.
Patients with additional orthopedic or medical pathology or disability/narcotic issues making them unable or unwilling to fill out follow-up forms specific to their artificial disc replacement occurred in 29% (7 of 24) of the failure group. Despite the fact these patients were considered failures based on ODI and VAS they reported a 92% satisfaction rate with the artificial disc and would repeat the surgery for the same result.
Failure Due to Additional Orthopedic Pathology:
• 29% (7 out of 24 patients)
• These 7 patients were failures due to <50% improvement in O.D.I. yet 92% reported satisfaction with A.D.R. and would repeat surgery for same result.
The overall causes of clinical failure in the LADR patients is explained in Table 6.
Interestingly, ADR patients are either a clinical success at three-month follow-up or failure. Only five patients in the entire group of 91 patients went from a success to failure after three months. One patient had an infection 18 months after the initial artificial disc replacement and four patients developed additional pathology unrelated to their artificial disc replacement. These patients did not have additional surgery but were considered failures due to the ODI scores. Only one patient went from failure to success after facet rhizotomy one year after artificial disc replacement.
Results of BMC Patients
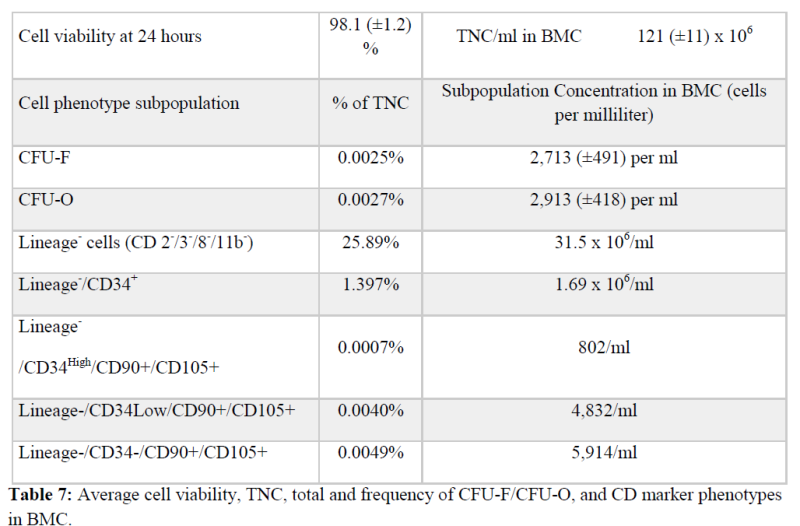
The average TNC concentration was 130 million per ml, the average mesenchymal (CFU-F) cell concentration was 2,702 per ml, and the average hematopoietic-endothelial progenitor (CD34+/Lineage–) concentration was 1.66 million per ml. The overall cell count data is explained in Table 7.
Adverse Events
There were no serious complications from harvesting the bone marrow concentrate or the disc injections. The most common events were transient pain at the harvest site and discomfort at the injection site, both of which typically resolved within 48 hours of treatment. Not every patient improved significantly, but no patient reported increases in ODI or VAS from pretreatment scores.
Progression to Surgery, Re-injection, and Correlations with BMC Cellularity
All patients were referred to this study after seeking a surgical consult for either an artificial disc replacement or fusion. After their BMC injection, two patients elected to have a second injection of BMC and two other patients had a surgical procedure within one- year (7 and 11 months), three patients elected to have surgery between one and two years (18, 22, and 24 months) years for a total of five patients through two years. There were no statistical differences between survivors and surgery dropouts in age, gender, BMI, number of levels injected, or cellularity in the BMC.
Outcome Assessment and Analysis
There were no serious complications from harvesting the bone marrow concentrate or the disc injections. The most common events were transient pain at the harvest site and discomfort at the injection site, both of which typically resolved within 48 hours of treatment. Not every patient improved significantly, but no patient reported increases in ODI or VAS from pretreatment scores.
Five patients in the BMC study had surgery within two years of the BMC injection and would be considered failures (19%). The clinical results on the remaining non-surgical patients are explained in figure 1. The average patient improved 64% in ODI and 72% in VAS. (Figure-1)
Discussion
This study is a prospective non-randomized IRB evaluation of 26 patients having a BMC disc injection compared to three FDA IDE studies involving the Maverick (25), Charite (31) and Kineflex (35) patients. The purpose of the study was to obtain preliminary data on the safety and efficacy of utilizing BMC to treat discogenic back pain compared to LADR surgery. The BMC study included injections into one (13 patients) or two discs (13 patients). The LADR were all one level surgeries.
The results in this group of 26 patients undergoing a single injection of BMC into 1 to 2 discs in the lumbar spine was unexpected. The two-year follow-up showed an average improvement in ODI of 64% and VAS of 72% (p<0.001). No patient was made worse, and only five patients underwent surgery during the follow-up (19%). These results are better than LADR. This conclusion is based on the morbidity of the surgery [several months recovery], the re-operation rate (5 %) and clinical failure rate (26%) compared to a BMC injection. The cost of the BMC procedure was $5000 compared to an average of $50,000 for the LADR.
The intervertebral disc [IVD] has very limited vascularity and capacity for self-repair. Current non-surgical and surgical treatment modalities for chronic discogenic back pain are aimed at symptomatic relief not disc repair. The use of mesenchymal stem cells from bone marrow concentrate to slow or reverse the degeneration of discs has been reported to yield promising results. Bone marrow derived mesenchymal stem cells are capable of stimulating both native disc cells and differentiating into IVD-nucleus pulposus cells [4,6,7,8,9,11,29,30].
Several human studies have recently been published documenting the safety and efficacy of utilizing biologics to treat chronic discogenic pain [8,9,10,21,31]. The injection of BMC into degenerated discs has been reported to be associated with increased water content within the IVD in about 40% of patients [1].
Limitations of the BMC study include no randomized control, no follow-up MRI scan data past one year. Follow-up was obtained in 100% of the BMC patients. The demographics of the patients in this study were similar to the FDA studies. The FDA studies were all one level surgeries.
Conclusion
Patients with chronic severe discogenic back pain have two surgical treatment options, fusion or LADR. There is minimal literature reporting the long-term efficacy of any non-operative treatment. Two-year follow-up data in treating single or two level lumbar pain with the BMC showed an improvement in ODI of 64% (p<0.001) and VAS of 72% (p<0.001). No patient was made worse by the procedure and there were no complications from the percutaneous injection of BMC into the disc. The BMC procedure took 30 minutes with no recovery time. Five BMC patients had surgery (19%). Utilizing MSCs derived from BMC, based on these preliminary results, may offer patients with single or two level discogenic lumbar pain a viable treatment option compared to LADR.
Acknowledgments
I would like to acknowledge the help of Dylan Meridith and Nick Collins in obtaining patient follow up.
Funding: There was no outside funding for this research.
Conflict of interest: No conflict of interest with the authors and this research.
References
1. Pettine K, Murphy M, Suzuki R, Sand T. Percutaneous Injection of Autologous Bone Marrow Concentrate Cells Significantly Reduces Lumbar Discogenic Pain Through 12 Months. Stem Cells. 2015;33: 146-156.
2. Pettine K, Suzuki R, Sand T, Murphy M. Treatment of discogenic back pain with autologous bone marrow concentrate injection with minimum two-year follow-up. Int. Orthop. 2016; 40:135-140.
3. Pettine K, Suzuki R, Sand T, Murphy M. Autologous Bone Marrow Concentrate Intradiscal Injection for the Treatment of Degenerative Disc Disease with Three-Year Follow-Up. IntOrthop 2017; Oct; 41(10):2097-2103. Doi: 10.1007/s00264-017-3560-9.
4. Gruber HE, Ingram JA, Davis DE, Hanley EN Jr. Increased cell senescence is associated with decreased cell proliferation in vivo in the degenerating human annulus. Spine J. 2006; 9: 210-215.
5. Meisel HJ, Ganey T, Hutton WC, Libera J, Minkus Y, et al. Clinical experience in cell-based therapeutics: intervention and outcome. Eur Spine J. 2006; 15 Suppl 3: S397-405.s
6. Zhang YG, Guo X, Xu P, et al. Bone mesenchymal stem cells transplanted into rabbit intervertebral discs can increase proteoglycans. ClinOrthopRelat Res. 2005; 430: 219-226.
7. Sakai D, Mochida J, Iwashina T, et al. Differentiation of mesenchymal stem cells transplanted to a rabbit degenerative disc model: potential and limitations for stem cell therapy in disc regeneration. Spine. 2005; 30: 2379-2387.
8. Sakai D, Mochida J, Iwashina T, et al. Regenerative effects of transplanting mesenchymal stem cells embedded in collagen to the degenerated intervertebral disc. Biomaterials. 2006; 27: 335-345.
9. Sheikh H, Zakharian K, De La Torre RP, Facek C, Vasquez A, et al. In vivo intervertebral disc regeneration using stem cell-derived chondroprogenitors. J Neurosurg Spine. 2009; 10: 265272.
10. Caplan AI, Correa D. The MSC: an injury drugstore. Cell Stem Cell. 2011; 9: 11-15.
11. Coric D, Pettine KA, Sumich A, et al. Prospective study of disc repair with allogenic chondrocytes presented at the 2012 Joint Spine Section Meeting. J Neurosurg Spine. 2013; 18: 85-95.
12. Rohlmann A, Mann A, Zander T, et al. Effect of an artificial disc on lumbar spine biomechanics: a probabilistic finite element study. Eur Spine J. 2009; 18:89-97.
13. Freeman BJC, Davenport J. total disc replacement in the lumbar spine: a systematic review of the literature. Eur Spine J. 2006; 15(Suppl 3): S439-47.
14. German JW, Foley KT. Disc arthroplasty in the management of the painful lumbar motion segment. Spine. 2005; 30(16S):S60-7.
15. Guyer RD, McAfee PC, Hochschuler SH, et al. Prospective randomized study of the Charité artificial disc: data from two investigational centers. Spine J. 2004 Nov-Dec; 4(6 Suppl):252S-9S.
16. Guyer RD, McAfee PC, Banco RJ, et al. Prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of lumbar total disc replacement with the Charite artificial disc versus lumbar fusion: Five-year follow-up. Spine J. 2009; Sept 18.
17. Blumenthal S, McAfee PC, Guyer RD, et al. A prospective, randomized, multicenter Food and Drug Administration investigational device exemptions study of lumbar total disc replacement with the Charité™ artificial disc versus lumbar fusion. Part I: evaluation of clinical outcomes. Spine. 2005; 30(14):1565-75.
18. Blumenthal SL, Ohnmeiss DD, Guyer RD, et al. Prospective study evaluating total disc replacement: preliminary results. J Spinal Disord Tech. 2003 Oct; 16(5):450-4.
19. Delamarter RB, Bae HW, Pradhan BB. Clinical results of ProDisc-II lumbar total disc replacement: report from the United States Clinical Trial. OrthopClin N Am. 2005; 36: 301-13.
20. Delamarter RB, Fribourg DM, Kanim LEA, et al. ProDisc artificial total disc replacement: Introduction and early results from the United States Clinical Trial. Spine. 2003; 28(20S): S167- 75.
21. Le Huec [a] JC, Basso Y, Aunoble S, et al. Influence of Facet and Posterior Muscle Degeneration on Clinical Results of Lumbar Total Disc Replacement: Two-Year Follow-Up. J Spinal Disord Tech. 2005 Jun; 18(3):219-23.
22. Panjabi M, Malcomson G, Teng E, et al. Testing of lumbar CHARITÉ discs versus fusions. Spine. 2007; 32(9):959-66.
23. Siepe CJ, Zelenkov P, Sauri-Barraza JC, et al. The fate of facet joint and adjacent level disc degeneration following total lumbar disc replacement: a prospective clinical, x-ray, and magnetic resonance imaging investigation. Spine. (Phila Pa 1976) 2010 Oct 15;35(22):1991-2003.
24. Zigler J, Delamarter R, Spivak JM, et al. Results of the prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of the ProDisc-L® total disc replacement versus circumferential fusion for the treatment of 1-level degenerative disc disease. Spine. 2007; 32(11):1155-62.
25. Park CK, Ryu KS, Jee WH. Degenerative changes of discs and facet joints in lumbar total disc replacement using ProDisc II: minimum two-year follow-up. Spine 2008 Jul 15;33(16):1755-61.
26. Shim CS, Lee S-H, Shin H-D, et al. Charité versus ProDisc: A comparative study of a minimum 3-year follow-up. Spine. 2007; 32(9):1012-8.
27. Kosharskyy B, Rozen D. Discogenic low back pain. Minimally invasive interventional therapies. AnasthesiolIntensivmedNotfallmedSchmerzther. 2007; 42(4):262-7.
28. Glassman S, Gornet MF, Branch C, et al. MOS short form 36 and oswestry disability index outcomes in lumbar fusion: A multicenter experience. Spine Journal. 2006; 6(1):21-6.
29. Wang Z, Perez-Terzic C, Smith J, et al. Efficacy of intervertebral disc regeneration with stem cells - A systematic review and meta-analysis of animal-controlled trials. GENE. 2015; 564: 1-8.
30. Jeffrey Zeckser, Michael Wolff, Jason Tucker, and Josh Goodwin, 'Multipotent Mesenchymal Stem Cell Treatment for Discogenic Low Back Pain and Disc Degeneration,' Stem Cells International. 2016; Article ID 3908389, 13 pages, 2016. https://doi.org/10.1155/2016/3908389
31. Pettine K, Dordevic M, Reducing Lumbar Discogenic Back Pain and Disability with Intradiscaal Injection of Bone Marrow Concentrate: 5-Year Follow-up. American Journal of Stem Cell Research. 2018; 2(1):1-4.
© 2018 medicalpressopenaccess.com. All rights reserved.