Overview on the Role of Breast Conservative Treatments for Early Breast Cancer with Low Risk of Recurrence: History, Standard and Controversies
Fabbri Nicolò Bagolini F Koleva Radica M Carcoforo P
DOI : https://doi.org/10.31546/IJCG.1001
Download Article : Peer-reviewed Article PDF
Abstract
Background: breast-conserving therapy is the mainstay for early-stage breast cancer, but this concept required a long process because comprehension of tumor biology was first necessary. In the last century several studies conducted to a progressive limitation of surgery and a progressive increase of complementary therapy, thanks to the early diagnosis of the screening in high-risk groups. The radiotherapy of the whole breast remains the gold standard after quadrantectomy, but new technical devices are leading to a modification of this approach. The advantage of breast conservative therapy is a conserved quality of life, but principal questions remain on the long term effects of these therapies and the risk of recurrences.Methods: We conducted an overview on the breast-conserving therapy for early-stage breast cancer with low risk of recurrences searching articles in the Pubmed literature with care on the role of intraoperative radiotherapy.
Conclusions: nowadays it become important to establish a correct therapeutic timing, adapting it to the characteristics of the individual patient and to the biology of the tumor. Radiotherapy and chemotherapy programs should be able to embrace according to modulable times and take into account the differences of each individual case. The reduction of post actinic toxicity represents an objective to be pursued, also for the possible overlap with the toxicity of the chemotherapeutic treatment and in this a fundamental role could be played by IORT. Moreover, considering the increase in patient survival that will inevitably lead to an increase in local recurrences, the possibility of a re-irradiation will be more and more frequent. Recent studies on the modulation of inflammation factors induced by the use of drugs during surgery could lead to new considerations on chemotherapy treatment. In conclusion, It could be useful to limit the field of action of complementary treatments for selected patients, like in the past it was proposed for surgical treatment, to allow any future therapy or reinterventions in case of recurrences, even with reconstructive intent.
Keywords: Intraoperative radiotherapy (IORT), breast conservative surgery (BCS), accelerated partial breast irradiation (APBI).
The History
One third of breast cancer cases occurs in patients over the age of 65 years, and in more developed countries this proportion rises to more than 47% after 70 years according to the updated Surveillance, Epidemiology, and End Results (SEER) database [1].
Radical mastectomy was introduced in 1882 by American surgeon William S. Halsted of Johns Hopkins. The Halsted operation consists in cutting out the breast, axillary lymph nodes and chest muscles. Before the National Cancer Institute's (NCI) initiation of the first controlled study in 1971, no comparative findings existed. Reported high rates of multifocality of breast cancer, based on pathologic studies, have been for many years a major deterrent to attempts to treat breast cancer with less mutilating surgical procedures than radical or total mastectomy [2].
The NCI study, directed by surgeon Bernard Fisher, has shown that the Halsted operation achieves no higher cure rate than the simple mastectomy that spares the muscles and uninvolved lymph nodes. Surgeon Alfred Meyer confirmed this conclusion in the first large-scale (1,686 cases) restrospective study.
Globally, breast cancer incidence rates are highest in North America and northern Europe. Breast cancer mortality rates have declined since 1975, attributed to the increased use of screening mammography and greater use of adjuvant treatments, including radiotherapy [3].
Breast conservation by irradiation as primary treatment for early breast cancer was initiated soon after the introduction of radiotherapy. In the 1920s large series of patients were treated with various techniques of irradiation, with or without preceding tumourectomy [4]. Keynes reported one of the first experience of Radium treatment of primary breast carcinoma in 1924. Results were instructive if unsatisfactory, for it was shown that a lump could be vanished with by local treatment with Radium, although it hardly ever prolonged the patient’s life. At the beginning of the last century mortality and morbility during and after surgey were high and the insertion of radium needles was a trivial operation compared with Halstead’s operation. In favourable cases an apparent cure could be affected without no mutilation whatsoever.Finally Author suggested that the lymphatic drainage could be dealt with more extensively with Radium than by dissection, since it was possible to treat effectively not only the axilla, but also the supra- and infra-clavicular regions and the intercostal spaces [5].
Subsequently the radiosensitivity of breast tissue has been shown by studies of the incidence of breast cancer in atom bomb survivors, and women subjected to multiple fluoroscopies or radiotherapy(6). From 1954 to 1966, 28 patients with small, well-outlined breast cancer were treated with a radical partial mastectomy followed by low-dose orthovoltage irradiation.In theese period many surgeons considered the replacement of any part of the operation by radiotherapy as a violation of the basic principles of cancer treatment and a true leap backwards.
In the 1950s some authors suggested that limited breast surgery followed by the postoperative radiotherapy regimen which was standard at that time, might be appropriate for a small subgroup of patients. However, an alarming number of local recurrences in this and other series led to abandon this method in 1961 and breast-conserving therapy (BCT) was no longer offered as an option, because of the number of breast cancer recurrences that had occurred. The disappointing results in the first 28 patients, 12 of whom developed a breast recurrence, were considered to constitute an argument against partial conservation of the breast for a long time. However, during a conference in Strasbourg in 1972, several French centers presented results of breast-conserving therapy (BCT), changing the previous opinions.
The fact that most recurrences were located near the excision site suggests that most of the breast recurrences originated from a residual tumor or in a premalignant [4] area but some authors considered that rarely a late breast cancer recurrences could be related to the tumourigenic effect of the irradiation.
The extensiveness of the local surgical therapy does not significantly alter rates of distant recurrence of disease or patient survival. The results of the randomized controlled studies of Atkins et al. and Veronesi and associates comparing Halsted mastectomy with wide local excision or quadrantectomy followed by local radiotherapy, supported these conclusions, at least for patient groups with T 1 N0 tumors [1].
Over the past 40 years, results from trials of the National Surgical Adjuvant Breast and Bowel Project (NSABP) have contributed significantly in reducing the extent of surgical procedures and in improving the outcome of patients with early-stage breast cancer [2]. In 1985 a milestone work by Holland allowed measuring the volume of the breast that was most exposed to the risk of residual cancer [1].
Holland indicated, in cases of breast cancer up to 2 cm in diameter, an area of higher risk of residual cancer around the main tumor which is, on average, the size of a whole breast quadrant. (about 9-10 cm in diameter).
Lastly, the NSABP B-06 trial compared lumpectomy and axillary node dissection with or without breast radiation with modified radical mastectomy in patients with tumors 4 cm or less in their greatest diameter. Similar to prior reports, the last update from that trial continues to demonstrate the value of lumpectomy and breast radiation as the preferred treatment in the majority of patients with invasive operable breast cancer as shown by data at 20 years of follow up [2].
The development of conservative surgery and complementary radiotherapy
In the early 90s, breast-conserving therapy for breast cancer became more widely used after randomized studies had shown that breast conservation plus radiotherapy and different forms of mastectomy resulted in equivalent rates of distant recurrence and survival [3].
According to the Geneva Tumour Registry, the proportion of all curative breast operations that were conservative rose from 3% before 1985, to 51% in 1990, then to 67% since 1998 [4]. One of the unresolved questions following disclosure of the results from the NSABP B-06 trial, as well as the Milan trial, was whether all patients with invasive breast cancer undergoing lumpectomy needed postoperative radiotherapy. It was hypothesized that patients with small tumors (≤1 cm) could potentially be spared from radiotherapy because they have lower rates of local recurrence [2]. In 1990 The Uppsala-Orebro Breast Cancer Study Group conducted a Randomized Trial to compare sector resection With or Without Postoperative Radiotherapy for Stage I Breast Cancer. In this trial, 381 women with invasive breast cancer had sector resection with meticulous control for complete excision of local disease plus axillary dissection.
187 women were randomly assigned to receive postoperative radiotherapy to a total mean dose of 54 Gy to the breast and 194 women to receive no further treatment. The local recurrence rate after 3 years was 2.9% in the group treated with radiation and 7.6% in the group treated only with the surgery but these groups did not differ significantly in overall survival. The author concluded that radiation therapy saves some patients from local recurrence but failed to indicate a group of patients that could avoid radiotherapy [3].
Modern external beam radiotherapy has low side-effects [5] (fatigue, local inflammation and breast oedema) that resolve rapidly in most patients, but since breast cancer is common, the absolute number of women with complication and side-effects is large.
After breast-conserving surgery, breast and chest wall pain are more frequent in irradiated than in non-irradiated patients, but this difference disappears after 6–18 months [4]. Holland postulated that if tumors measuring 2 cm or less were removed with a margin of 4 cm, which is, on average, equal to a whole breast quadrant, about 5% of the patients would harbor invasive tumor foci in the remaining breast. These foci could theoretically be the sources of short-term recurrence. Moreover, if radiation were less effective on non-invasive cancers than on invasive tumor, the expected rate of recurrences, presumably long-term, would be significantly higher after a limited excision than after a total quadrantectomy. However, the effectiveness of postoperative radiation therapy in preventing or delaying local recurrence has, in general, been satisfactorily documented [1]. Sarcomas appearing in the breast or the chest wall are considered radiation induced , but fortunately they are rare, concerning less than 0.5% of irradiated patients [4].Compared to squamous cell carcinoma, breast cancer seems to show a different sensitivity towards higher single doses [6]. In 1989, Fowler postulated an alpha/beta ratio of 4 for breast cancer as its best approximation instead of 10 for most SCC. This value was strongly supported by the clinical outcome of Canadian and British ypofractionation Trials [7].
Recent meta-analyses of randomised trials have established that breast cancer mortality can be significantly reduced by loco-regional radiotherapy (RT), and that the increased intercurrent mortality observed in older trials was caused by an excess in cardiovascular deaths, presumably avoidable, associated with the earlier techniques [4]. We found meta-analyses of randomised trials have established that breast cancer mortality can be significantly reduced by loco-regional radiotherapy (RT), and that the increased intercurrent mortality observed in older trials was caused by an excess in cardiovascular deaths, presumably avoidable, associated with the earlier techniques. In a meta-analysis of individual patient data for 10 801 women in 17 randomized trials of radiotherapy versus no radiotherapy after breast-conserving surgery, data suggest that radiotherapy reduced the 10-year risk of any (ie, locoregional or distant) first recurrence from 35.0% to 19.3% (absolute reduction 15.7%, 95% CI 13.7–17.7, 2p<0.00001) and reduced the 15-year risk of breast cancer death from 25.2% to 21.4%. Authors conclusions were that for many women with early-stage breast cancer, breast-conserving surgery can remove any macroscopic disease that has been detected; however, some microscopic tumor foci might remain in the conserved breast that could, if untreated, lead to locoregional recurrence or life-threatening distant metastases, or both.
Both proportional and absolute reductions in the annual recurrence rate are largest in the first year but the recurrence rate continues to be somewhat lower throughout the first decade, whereas the reduction in
breast cancer death rate becomes definite only after the first few years and appears to continue into the second decade(8). Moreover, the absolute benefit from boost radiation treatment is particularly marked in young patients and is indicated for all patients less than 50 years of age. In older patients, the omission of boost irradiation may be considered in the absence of risk factors for local recurrence [4]. However patients with tumor-involved margins are recommended to undergo re-excision to tumor-free margins or mastectomy [9]. the overall survival and survival of patients with T1N0 breast cancer are now nearly identical to women without breast cancer [10].
The role of Tamoxifen
The principal objectives of the B-21 study by the National Surgical Adjuvant Breast and Bowel Project (NSABP) were to examine whether Tamoxifen was as effective as radiation in controlling ipsilateral breast tumor recurrence (IBTR) and whether the addition of Tamoxifen to radiation was superior to radiation alone in terms of local and systemic control of the disease.
The cumulative incidence of IBTR over an 8 year period was 16.5% with Tamoxifen alone, 9.3% with radiation and placebo, and 2.8% with radiation and tamoxifen. Thus, this trial demonstrated that in the group of node-negative patients with small invasive tumors treated by lumpectomy, Tamoxifen was not as effective as breast radiation in controlling the disease in the breast. It further demonstrated that the combination of Tamoxifen and breast radiation resulted in better local control of the disease in the breast than either modality alone [2]. With a median of 11.2 years from the current trial the long-term results of follow-up published in 2007 continue to support the need for local breast irradiation and adjuvant therapy in the management of patients with these small cancers [11].
The sentinel Node concept
Since to the early ’90s, conservative surgery has also investigated the aspect of lymph nodal axillary dissection as a standard treatment. It was known that in the majority of patients with operable breast cancer (about 75%) the axillary nodes were found to be histologically negative at the time of surgery. These patients do not derive any therapeutic benefit from the axillary dissection but could experience significant morbidity as a result of the procedure [6].
In the early 1990s, Morton et al. studied the role of blue dye injection into the skin around the site of the primary melanoma to identify and remove the regional lymph node (SN) and examine it for metastatic involvement and avoid an unnecessary radical lymphadenectomy. The Sentinel Node Biopsy concept was soon extended to breast cancer to predict axillary status in breast cancer by Krag, Giuliano, and the Milan group. Studies conducted in the 90’s by Prof. Veronesi indicated that SNB makes it possible avoid Axillary Dissection in the considerable proportion of breast cancer patients with an uninvolved SN, thereby reducing postoperative morbidity and the length of postoperative hospitalization [12].
IORT (Intraoperative Radiotherapy)
When breast conserving surgery (BCS) is chosen, excision is commonly followed by 5 weeks of whole breast irradiation (WBI), with or without a boost to the tumor bed. Long radiation schedules are a burden for many women [13].
This has stimulated an interest in accelerated partial breast irradiation (APBI) that can reduce overall treatment time without compromising oncological outcomes or cosmesis. Intraoperative radiation therapy (IORT) is an attractive APBI approach because it delivers the entire radiation treatment during surgery [13].
IOERT (intraoperative electron radiotherapy) in breast cancer is used either as a boost (10–12 Gy) followed by whole breast irradiation (WBI) or as full-dose partial breast irradiation (PBI, 20–24 Gy) during breast-conserving surgery(19). The method was originally introduced by the Medical College of Ohio (MCO) in Toledo, Ohio, USA, and the Centre Regional de Lutte Contre Le Cancer (CRLC) in Montpellier, France, based on reports of 72 patients treated with an electron boost (intraoperative electron radiotherapy (IOERT) [1].
The most frequently reported boost techniques comprise external electrons or photons with fractionated doses of 10–16 Gy, or interstitial implants as high dose rate brachytherapy. In contrast to postoperative irradiation, intraoperative radiotherapy (IORT) offers the possibility to apply a high single dose at the time of BCS with utmost precision due to direct visualization [19].
The relative distribution of tumor foci around the reference tumor seems independent of reference tumor size although a linkage to the distance to the reference tumor seems to exist. In 1985 Holland indicated a risk of residual tumor foci of about 5% after full quadrantectomy [2]. Pathological analyses revealed that the greatest tumour cell density (up to 90% of microscopic remainders) is observed in an area of 4 cm surrounding the macroscopic tumour edge [1].
IORT has some advantages:
- Geographic misses are avoided due to direct visualization of the tumor bed;
- A high single dose is delivered with utmost precision to small volumes, completely sparing the skin and ensuring a good long-term cosmetic outcome;
- High single doses seem to induce biological mechanisms with verifiable antitumor capability in in-vitro cell-line studies. In addition, IOERT markedly shortens the overall treatment time.
Even in high-risk groups like triple-negative or locally advanced breast cancers, IOERT contributes to long-term local control rates of more than 90%. For selected low-risk groups, IOERT as PBI with 21 Gy seems to be a viable treatment alternative to standard WBI [19].
Two randomized controlled trials, TARGIT-A (used 50-kV X-rays) and ELIOT (used electrons), have shown that IORT is non-inferior to EBRT in terms of LRR (Local Recurrence Rate) when delivered to patients with early breast cancers and specific tumor characteristics [20].
But restriction of radiation therapy to the tumor bed during surgery might be adequate only for selected patients? [10]
The ELIOT Study
Intraoperative radiotherapy (IORT) can be used to deliver accelerated partial breast irradiation (APBI) in a single fraction at the time of tumor excision. The largest experience with this technique is from the Milan group where Veronesi and colleagues first perform a standard quadrantectomy. During surgery, the breast parenchyma is reapproximated using sutures, and IORT is delivered with a mobile linear accelerator. Originally used to deliver the tumor bed ‘‘boost,’’ the Milan group has subsequently extended the use of IORT to the sole modality for small, low-risk tumors [9].
In a series of 1305 unselected patients treated with single electron dose (intraoperative electron radiotherapy (IOERT), Veronesi et al. identified an excess of ipsilateral breast recurrence tumor (IBRT) in the intraoperative radiotherapy group compared with the external radiotherapy group, in the quadrant initially affected by the disease (2.5% vs 0.4% at 5 years), and as new tumours in the other quadrants of the same breast (1.9% vs 0% at 5 years), with a percentage of 5-year occurrence of IBTR in the intraoperative radiotherapy group of 4.4% (however, lower than maximal prediction of 7.5%) [8].

In ELIOT study overall survival at 5 years was identical, 96.8 % for the ELIOT group vs 96.9 % for the EBRT group. The 10-year survival remained similar (89.8 % for ELIOT and 92.0 % for EBRT patients).
Overall, ELIOT patients had a higher 5-year recurrence rate than EBRT patients (4.4 % vs 0.4 %, p = .0001). However, ELIOT patients can be divided into low- and high-risk groups based on tumor size, receptor status, nodal positivity, and grade. ELIOT Low-Risk women (69.4 % of the ELIOT patients) had a 5-year IBTR rate of only 1.5 % compared with 11.3 % for the 30.6 % of ELIOT patients with 1 or more high-risk factors [13].
In ELIOT trial postoperative discovery of predefined factors (eg, lobular carcinoma) could trigger the addition of external beam radiotherapy to targeted intraoperative radiotherapy (in an expected 15% of patients) [5].
Some authors have investigated the possibility of anticipating intraoperative radiotherapy on tumor tissue before quadrantectomy. The advantage in this case could be greater precision in radiation delivery. In 2011 Kimple et al. published results of theyr study about local control following single-dose intraoperative radiotherapy prior to surgical excision in early-stage breast cancer.
Between March 2003 and July 2007, 71 patients underwent IORT: IORT alone was given in 53 patients, 11 received whole breast radiotherapy, and 7 underwent a mastectomy. It was used a dose of 15 Gy based on a calculation of a biologically equivalent dose. Four women experienced invasive ipsilateral breast failures (1 new primary, 3 margin recurrences) and the 3-year actuarial local control of 92% was lower than anticipated.
With an 8% of three-year in breast recurrence authors concluded that intraoperative radiotherapy delivered to an in situ tumor is feasible, but the local control rate at 3.5 years is concerning [9].
The TARGIT study
The TARGIT-A Trial randomized 3,451 patients either to standard EBRT treatment or to TARGIT-A.
Eligibility criteria were age C 45 years, tumor size B 3.5 cm, N0–1, M0, and unifocal invasive ductal carcinoma. If the participating institution determined the patient was at high risk for recurrence, an additional 5 weeks of EBRT was given, calling this
‘‘risk adapted IORT.’’ The Trial began in March 2000 [10].
In the TARGIT study in selected patients with early breast cancer, one dose of x-ray intraoperative irradiation resulted in much the same proportions of patients having local recurrence as with conventional radiotherapy at 4 years (1.20% vs 0.95%), but by 5 years, local recurrence was signifi cantly greater in the TARGIT group (3.3% vs 1.3%; p=0.042) [8].

IORT in elderly patients
Nowadays in women aged 70 and over, diagnosis of early breast cancer with screen-detected breast lead to a higher proportion of breast-conserving surgery.
However elderly patients often have significant co-morbidities or serious difficulties preventing them from attending a radiation daily treatment. Because of this, elderly patients often decline RT or even opt for a mastectomy to avoid radiation treatment.
The German TARGIT-E trial was launched to demonstrate that elderly patients, who are often undertreated as they often do not comply with standard 3–6 weeks of EBRT, should be treated at the time of surgery with IORT when they present with small breast cancer.
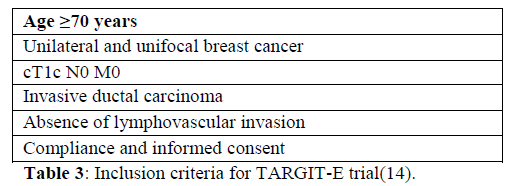
The authors concluded that whether radiotherapy is beneficial in the elderly still remains a matter of debate. IORT along with BCS in a selected subgroup of elderly patients (> 70years, pT1N0 tumors) could represent a valid option for better local control.
Effect in wound fluid
The initial TARGIT-A publication did not differentiate between pre pathology and post pathology patients or Targit boost patients. The TARGIT update shows these strata are not equivalent, with post pathology having higher local recurrence rates than pre pathology.
The authors attribute the difference either to delay in wound fluid suppression of tumor cells since there is a delay of radiation in post pathology TARGIT,or to a geometric miss when inserting the applicator postsurgery [10].
A supposed ‘biological’ advantage of IORT is an immediate effect of irradiation on the tumor microenvironment by abrogation of the proliferative cascade induced by wound healing after surgery. In vitro, wound fluid has been described to stimulate tumor cell proliferation and invasion, which can be blocked by high-dose IORT.
Fabris et al. demonstrated that IORT elicits effects that go beyond the direct killing of residual tumor cells with a different wound response, inducing the expression of miR-223 in the peritumoral breast tissue. miR-223 downregulated the local expression of epidermal growth factor (EGF), leading to decreased activation of the EGF receptor (EGFR) on target cells.
Intraoperative RT induces miR-223 expression in peri-tumoral breast tissue miR-223 directly regulates the expression of epidermal growth factor.
Authors suggest that proper selection of the 'right' drug for the 'right' patient will not suffice to obtain the most effective treatment for each BC patient. The accurate choice of the most appropriate window of time for drug administration during the course of the disease will also prove crucial for the success of the therapy [15].
IORT and cosmesis
One of the most frequent complications in breast conservative surgery is the wound seroma. In ELIOT study seroma needing more than three aspirations were more frequent in the targeted intraoperative radiotherapy group than in the external beam radiotherapy (EBRT) group [5].
However compared with the conventional arm, ELIOT reported less skin damage (i.e., erythema, dryness, hyper-pigmentation, or itching), and no differences for fibrosis, retraction, pain or burning, but a higher incidence of radiologically determined fat necrosis,
(5 % in ELIOT trial, versus 2 % than EBRT). In addition, ELIOT had less pulmonary toxicity than the EBRT as diagnosed by follow-up spiral CT [13]. In TARGIT A trial wound complications were similar between groups, but grade 3 or 4 skin complications were significantly reduced with TARGIT vs EBRT [16].
Future perspectives
Other aspects of breast-conserving surgery must be investigated.
IART®: Intra-Operative Avidination for Radionuclide Therapy
In a recent pilot study published by Paganelli et al in 2010, on a sample of 35 patients, exclusive radiotherapy was located at the tumor bed in the immediate postoperative period using the chelating capacity of the avidin protein. (IART®: Intra-Operative Avidination for Radionuclide Therapy).
After tumor excision, the surgeon injected native avidin diluted in 30 ml of saline solution into and around the tumor bed.
This protein was applied by the surgeon with the aim of concentrating the maximum dose of the radioactive isotope in the days following the operation.
Between 12 to 24 h after surgery, 3.7 GBq 90Y-biotin spiked with 185 MBq 111In-biotin was administered intravenously. No side effects were observed after avidin administration and 90Y-biotin infusion [17].
IORT delivered with Xoft® Axxent® eBx™
Xoft® is a balloon catheter born for brachytherapy with single-entry, which can be inserted into the tumour cavity by the surgeon at the time of surgery or after. The Xoft® Axxent® eBx™ System has been used to treat early breast cancer with a multi fraction accelerated partial breast radiation.
The disadvantage of brachytherapy balloons is that the radiation treatment is not concluded at the time of the operation, but once the device has been placed, radiation is delivered in ten fractions twice a day over five consecutive days.
The purpose of the phase IV Xoft® Axxent® eBx™ IORT trial is to assess the safety and efficacy of the Xoft® Axxent® eBx™ System accrual goal of this trial is 1,200 patients and the first patient was recruited in 2012 [14].
Recently, research on the role of non-steroidal anti-inflammatory drugs in conservative surgery for breast cancer has appeared in the literature.
In a work published in 2019 by Dell’Omo et al. of the Milan group, the ability of NSAIDs to determine the inhibition of pro-inflammatory factors and the activation of the anti-oncogenic p53 / p21 pathway was studied. This study suggests a new strategy for the design of anticancer drugs, through the modulation of the pro-inflammatory cascade [18].
In a retrospective study published in 2018 Desmedt et al. show that the intra-operative administration of ketorolac during breast conservative surgery is statistically significantly associated with a reduction of distant recurrences in patients with early breast cancer and increased BMI [19].
Discussion
Based on the actual literature the absolute recurrence reduction produced by radiotherapy and the absolute recurrence risk remaining even with radiotherapy varies significantly with age,
tumor grade, ER status, and tamoxifen use, even after adjustment for all other factors. Tumour size is independently predictive of absolute recurrence risk although not of the absolute risk reduction [8]. In patients not receiving chemotherapy, it is recommended that RT should start within 8 weeks(4) however late problems in wound healing may rarely lead to a delay in the administration of complementary radiotherapy. In these cases, intraoperative radiotherapy would be useful in avoiding therapeutic delays.
Since newer adjuvant programs tend to call for increasing numbers of chemotherapy cycles, potentially detrimental delays of more than 6 months in starting RT may become frequent. Although the effectiveness of RT could be enhanced by concomitant chemotherapy, toxicity might also be increased. For this reason, the concomitant administration of anthracyclines and RT is not recommended.
To date, there is still no unanimity on the correct sequence of adjuvant chemotherapy and radiotherapy. When both chemotherapy and RT are to be given, the following sequence is possible: RT followed by chemotherapy, chemotherapy followed by RT, concurrent radio-chemotherapy, and RT ‘sandwiched’ between courses of chemotherapy [4].
Despite this optimism, caution should be expressed regarding the unknown consequences of combinations of regional RT with newer cardiotoxic chemotherapy agents. However, it is unclear whether or not delaying RT until after completion of chemotherapy decreases loco-regional control, and the survival consequences are unknown. Retrospective studies on the impact of delaying RT yielded conflicting results. In these cases exclusive intraoperative radiotherapy could avoid the alternation between radiotherapy and adjuvant chemotherapy, reducing the waiting time for chemotherapy treatment. Nonetheless, there is a current tendency to favor the early administration of chemotherapy, when indicated.
The ELIOT trial has contributed to our understanding of whether a single-dose treatment using electrons may be possible. The scientific community does not unanimously support the effectiveness of IORT and still raises concerns about introducing IORT as a standard treatment option for breast cancer [14].
It appears, however, that IOERT APBI may have a subset of low-risk women (ASTRO suitable, ELIOT Low Risk, Luminal A) for whom IOERT could be effective, with a recurrence rate in the 2 % range at 5 years.
Recently a single-center experience of the University of Verona reported only 1 recurrence at a mean follow-up of 46 months in 226 low-risk women treated with 21 Gy [13]. The inclusion of patients with higher risk profiles including triple-negative disease, ER-negative disease, young age, and large tumors ([2 cm) may be contraindicated. Recently published ASTRO consensus guidelines recommend cautious consideration of APBI for women less than 60 years old, tumors >2 cm and ER-negative disease. Another problem related to the increase in early diagnosis is an increase in breast-conserving surgery with a consequence: the increase of the demand for radiotherapy services required to treat breast cancer patients.
Intraoperative radiotherapy would make it possible to simplify the patients' treatment plan, lightening the workload of radiotherapy centers. Alternative approaches to treatment sequencing should, therefore, be investigated.
Early initiation of each treatment modality after surgery can be facilitated both by using alternating ‘sandwich’ schedules, or by the simultaneous administration of RT and chemotherapy, and may be beneficial regarding both loco-regional control and survival.
Furthermore, in consideration of the increase in overall survival of patients treated for early breast cancer and the highest risk of local recurrence if conservative surgery is performed without radiotherapy, it would be necessary to ask what damage the radiotherapy produces on the entire breast especially if putting at risk the healing of re-operations especially if reconstructive.
The increase in local recurrence does not seem to substantially change the overall survival of patients. Based on data in the literature, the ten-year survival of patients with locoregional recurrence is estimated to be over 80%.
These datas suggest the search for therapeutic solutions with the aim of maintaining a lifestyle that is as similar as possible to the starting one.
In selected situations, it is still possible to consider a second conservative surgery, particularly if the patient had not received radiation therapy on the breast volume. The diameter of the relapse (<2cm) and the interval time from the primary surgery (> 4 years) are the two factors that allow identifying women possibly candidates for a conservative re-intervention and with a good chance of success [16].
Close collaboration between breast and plastic surgeons and the oncologist is required in selecting patients for reconstruction, as the need for appropriate adjuvant radiation and systemic therapy should be the overriding concern. There are limited data defining and quantifying the effects of radiotherapy on the results of reconstructive surgery. Much of the data come from retrospectively analyzed surgical series. On the negative side, external boost treatment has been shown to significantly reduce the proportion of favorable long-term cosmetic results observed.
Telangiectasias can result from an excessive dose delivered to vessels that are located in the first 5 mm beneath the epidermis, and dose- and volume-dependent fibrosis can lead to breast retraction and deformity [4].
The use of exclusive IORT allows limiting actinic damage to the tumor bed only, maintaining an adequate level of safety on local long-term recurrences and allowing patients to have a total adhesion to radiotherapy treatment due to its uniqueness, overcoming the problems related to the logistics of radiotherapy centers and the comorbidities of patients who often do not allow the development of the entire radiotherapeutic plan.
However, the difficulty (with IOERT APBI) is not only to define patients at low risk of harboring microscopic disease beyond the tumor site but also to define the proper coverage of the tumor bed.
Krechetov estimates that to ensure uniform coverage of microscopic residual disease, the IOERT applicator should have a circumferential dimension at least 1.5 to 2 cm larger than the maximum tumor dimension(13).
The applicator sizes used in the ELIOT Trial are not specified, but the current guidelines for ELIOT at the EIO indicating larger field sizes, as suggested by Leonardi, are now preferred.
In Verona, where applicator size was selected to be approximately 2 cm circumferentially larger than the largest tumor dimension, the median applicator size was 6 cm [13].
The value of this study is that the treatment was relatively standardized during the years of this prospective study.
Current evidence demonstrates that IORT is ready for roll-out; it is time to let well-selected and informed patients be offered this treatment option in the current clinical practice [14].
Conclusion
Never before has it become important to establish a correct therapeutic timing, adapting it to the characteristics of the individual patient and to the biology of the tumor. Radiotherapy and chemotherapy programs should be able to embrace according to modulable times and take into account the differences of each individual case.
The reduction of post actinic toxicity represents an objective to be pursued, also for the possible overlap with the toxicity of the chemotherapeutic treatment and in this a fundamental role could be played by IORT. Moreover, considering the increase in patient survival that will inevitably lead to an increase in local recurrences, the possibility of a re-irradiation will be more and more frequent. Recent studies on the modulation of inflammation factors induced by the use of drugs during surgery could lead to new considerations on chemotherapy treatment.
In conclusion, It could be useful to limit the field of action of complementary treatments for selected patients, like in the past it was proposed for surgical treatment, to allow any future therapy or reinterventions in case of recurrences, even with reconstructive intent.
Acknowledgments: None.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
1. Holland R, Veling SHJ, Avunac M, Hendriks JHCL. Histologic multifocality of tis, T1–2 breast carcinomas implications for clinical trials of breastâ€conserving surgery. Cancer. 1985; 56:979-990.
2. Mamounas EP. NSABP breast cancer clinical trials: recent results and future directions. Clin Med Res. 2003; 1:309-326.
3. Liljegren G, et al. Sector resection with or without postoperative radiotherapy for stage I breast cancer: five-year results of a randomized trial. J Natl Cancer Inst. 1994 ; 86:717-722.
4. Kurtz J, EUSOMA Working Party. The curative role of radiotherapy in the treatment of operable breast cancer. Eur J Cancer. 2002; 38:1961-1974.
5. Vaidya JS, et al. Targeted intraoperative radiotherapy versus whole breast radiotherapy for breast cancer (TARGIT-A trial): An international, prospective, randomised, non-inferiority phase 3 trial. Lancet. 2010; 376:91-102.
6. Sedlmayer F, et al. Boost IORT in Breast Cancer: Body of Evidence. Int J Breast Cancer. 2014; 2014: 472516.
7. Sedlmayer F, et al. Intraoperative radiotherapy (IORT) as boost in breast cancer. Radiat Oncol. 2017; 12: 23.
8. Darby S, et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: Meta-analysis of individual patient data for 10 801 women in 17 randomised trials. Lancet. 2011; 378 :1707-1716.
9. Kimple RJ, et al. Local control following single-dose intraoperative radiotherapy prior to surgical excision of early-stage breast cancer. Ann Surg Oncol. 2011; 18:939-945
10. Silverstein MJ, et al. Intraoperative Radiation Therapy: A Critical Analysis of the ELIOT and TARGIT Trials. Part 2—TARGIT. Ann Surg Oncol. 2014; 21:3793–3799.
11. Fisher ER, et al. Pathobiology of small invasive breast cancers without metastases (T1a/b, N0, M0): National Surgical Adjuvant Breast and Bowel Project (NSABP) protocol B-21. Cancer. 2007; 110:1929-1936.
12. Veronesi U, et al. Sentinel lymph node biopsy in breast cancer: Ten-year results: Of a randomized controlled study. Ann Surg. 2010; 251:595-600.
13. Silverstein MJ, et al. Intraoperative Radiation Therapy: A Critical Analysis of the ELIOT and TARGIT Trials. Part 1-ELIOT. Ann Surg Oncol. Nov; 21: 3787-3792.
14. Esposito E, Douek M. Update on intraoperative radiotherapy: New challenges and issues. Ecancermedicalscience. 2018; 12:793.
15. Fabris L, et al. Radiotherapy-induced miR-223 prevents relapse of breast cancer by targeting the EGF pathway. Oncogene. 2016; 35:4914-4926.
16. Julia Kaisera, et al. Intraoperative Electron Radiotherapy (IOERT) in the Treatment of Primary Breast Cancer. Breast Care. 2018; 13:162-167.
17. Paganelli G, et al. IART® (Intra-Operative Avidination for Radionuclide Therapy) for accelerated radiotherapy in breast cancer patients. Technical aspects and preliminary results of a phase II study with 90Y-labelled biotin. Ecancermedicalscience. 2010; 4:166.
18. Dell’Omo G, et al. Inhibition of SIRT1 deacetylase and p53 activation uncouples the anti-inflammatory and chemopreventive actions of NSAIDs. Br J Cancer. 2019; 120:537-546.
19. Desmedt C, et al. Potential benefit of intra-operative administration of ketorolac on breast cancer recurrence according to the patient’s body mass index. J Natl Cancer Inst. 2018; 110:1115-1122.
© 2018 medicalpressopenaccess.com. All rights reserved.

